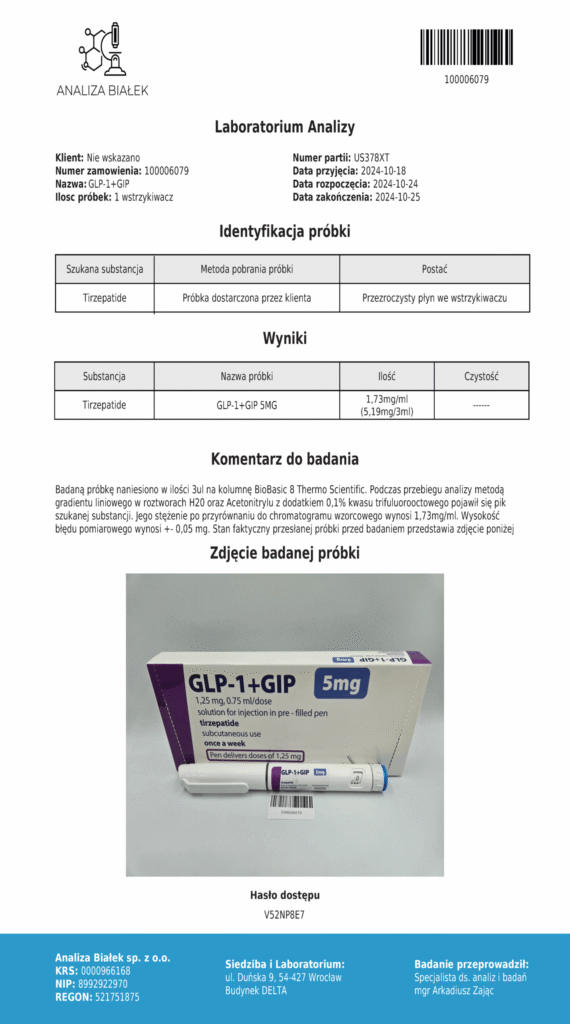
Tirzepatide pen 5 mg - characteristics of a dual GIP/GLP-1 agonist
Deadline tirzepatid pen refers to the dosage form used in clinical trials of a synthetic peptide analog with activity against GIPR and GLP-1R receptors. In Phase III study documentation, the term refers to a finished dosage form that allows subcutaneous administration once a week under controlled clinical protocols.
In a laboratory context, the term tirzepatid pen can function as a reference model for studying the pharmacokinetics of subcutaneous administration and assessing the dose-effect relationship for incretin agonists.
Mechanism of action of Tirzepatide pen in receptor models
Tirzepatide is a dual agonist of GIP and GLP-1 receptors, which belong to the G protein-coupled receptor (GPCR) family.
Molecular studies have shown:
activation of the cAMP pathway in pancreatic β-cells,
Increased glucose-dependent insulin secretion,
Suppression of glucagon secretion under hyperglycemic conditions,
Effects on β-arrestin signaling,
modulation of central pathways regulating appetite.
The mechanism of dual GIP/GLP-1 agonism was described in detail in a study by Min T. et al. (2023, PMC10107501), indicating a possible synergistic effect in the regulation of energy homeostasis.
Clinical data from the tirzepatide study
In the study SURMOUNT-1 (NEJM 2022; PubMed 35658024) analyzed doses of 5 mg, 10 mg and 15 mg administered once a week for 72 weeks.
For the dose corresponding to the form tirzepatid pen 5 mg reported:
Average weight reduction of ~15% from baseline,
Significant improvement in metabolic parameters,
reduction in waist circumference and triglycerides,
improving markers of insulin resistance.
In the program SURPASS-2 (NEJM 2021) showed a significant reduction in HbA1c and greater weight reduction compared to the selective GLP-1 agonist, the Semaglutide.
Online availability and where to buy tirzepatid pen
In the context of clinical trials, the term tirzepatid pen refers to the dosage form used in programs such as SURPASS and SURMOUNT. In the regulated market, the molecule Tirzepatide is a compound developed for therapeutic use and is subject to regulations applicable to medicinal products.
In the laboratory environment, analogs or reference materials based on this molecule can be offered by specialized research reagent suppliers as:
- GIPR and GLP-1R receptor research substances,
- Materials for pharmacodynamic and pharmacokinetic analyses,
- Analytical standards for GPCR signaling studies,
- Reagents for glucose and energy metabolism projects.
Online availability applies only to entities marketing chemical reagents or reference materials for research applications. In such cases, documentation should include:
- purity specifications (e.g., HPLC/UPLC),
- certificate of analysis (CoA),
- storage and stability conditions,
- Research Use Only (RUO) designation,
- batch identification data.
Dosage schedule in the study
The clinical protocols used an escalation scheme:
2.5 mg (introduction),
Then 5 mg,
With the possibility of increasing to 10 mg or 15 mg.
A dose of 5 mg, corresponding to the variant tirzepatid pen 5 mg, provided an important reference point in dose-effect and tolerance analyses.
Research relevance
In laboratory marketing, tirzepatyd functions as a reference material for research on:
Incretin receptor pharmacology,
GPCR signaling,
Energy balance regulation,
Glucose and lipid metabolism.
The formulation is intended for research and analytical applications only.